Learninsta presents the core concepts of Biology with high-quality research papers and topical review articles.
Applications In Medicine
Recombinant Human Insulin
The Human insulin is synthesized by the β cells of Islets of Langerhans in the pancreas. It is formed of 51 aminoacids which are arranged in two polypeptide chains, A and B. The polypeptide chain A has 21 amino acids while the polypeptide chain B has 30 amino acids. Both A and B chains are attached together by disulphide bonds.
Insulin controls the levels of glucose in blood. It facilitates the cellular uptake and utilization of glucose for the release of energy. Deficiency of insulin leads to diabetes mellitus which is characterized by increased blood glucose concentration and a complex of symptoms which may lead to death, if untreated. A continuous program of insulin dependence is required to treat this deficiency.
In the early years, insulin isolated and purified from the pancreas of pigs and cows was used to treat diabetic patients. Due to minor differences in the structure of the animal insulin as compared to human insulin, it resulted in the occurrence of allergic reactions in some diabetic patients.
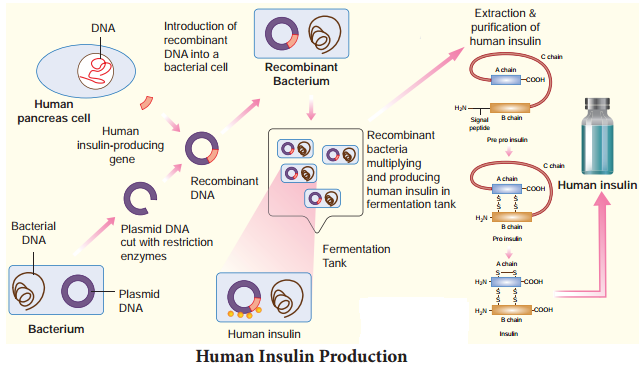
Production of insulin by recombinant DNA technology started in the late 1970s. This technique involved the insertion of human insulin gene on the plasmids of E.coli. The polypeptide chains are synthesized as a precursor called pre-pro insulin, which contains A and B segments linked by a third chain (C) and preceded by a leader sequence.
The leader sequence is removed after translation and the C chain is excised, leaving the A and B polypeptide chains (Fig. 9.1). Insulin was the first ever pharmaceutical product of recombinant DNA technology administered to humans. The approval to use recombinant insulin for diabetes mellitus was given in 1982. In 1986 human insulin was marketed under the trade name Humulin.
Interferons
Interferons are proteinaceous, antiviral, species specific substances produced by mammalian cells when infected with viruses. Interferons were discovered by Alick Isaacs and Jean Lindemann in 1957. Based on the structure of interferons they are classifid as α, β and γ interferons.
They stimulate the cellular DNA to produce antiviral enzymes which inhibit viral replication and protect the cells. Interferons could be isolated from blood, but the amount of blood required for isolation of interferons is enormous and not practical. To overcome this issue interferons could be produced by rDNA technology.
The yeast Saccharomyces cerevisiae is more suitable for production of recombinant interferons than E.coli, since E.coli does not possess the machinery for glycosylation of proteins. Interferons are used for the treatment of various diseases like cancer, AIDS, multiple sclerosis, hepatitis C and herpes zoster. In spite of the therapeutic applications interferons are not within the reach of the common man due to high cost for its production.
Recombinant Vaccines
Recombinant DNA technology has been used to produce new generation vaccines. The limitations of traditional vaccine production could be overcome by this approach. The recombinant vaccines are generally of uniform quality and produce less side effects as compared to the vaccines produced by conventional methods. Different types of recombinant vaccines include subunit recombinant vaccines, attenuated recombinant vaccines and DNA vaccines.
Subunit recombinant vaccines
Vaccines that use components of a pathogenic organism rather than the whole organism are called subunit vaccines; recombinant DNA technology is very suited for developing new subunit vaccines. It includes components like proteins, peptides and DNAs of pathogenic organisms. The advantages of these vaccines include their purity in preparation, stability and safe use.
Attenuated recombinant vaccines
This includes genetically modified pathogenic organisms (bacteria or viruses) that are made nonpathogenic and are used as vaccines. It is now possible to genetically engineer the organisms (bacteria or viruses) and use them as live vaccines and such vaccines are referred to as attenuated recombinant vaccines.
DNA Vaccines
Genetic immunisation by using DNA vaccines is a novel approach that came into being in 1990. The immune response of the body is stimulated by a DNA molecule. A DNA vaccine consists of a gene encoding an antigenic protein, inserted onto a plasmid, and then incorporated into the cells in a target animal. DNA instructs the cells to make antigenic molecules which are displayed on its surfaces.
This would evoke an antibody response to the free floating antigen secreted by the cells. The DNA vaccine cannot cause the disease as it contains only copies of a few of its genes. DNA vaccines are relatively easy and inexpensive to design and produce. Vaccines produced by these new techniques have definite advantages like producing target proteins, long lasting immunity and trigger immune response only against specific pathogens with less toxic effects.
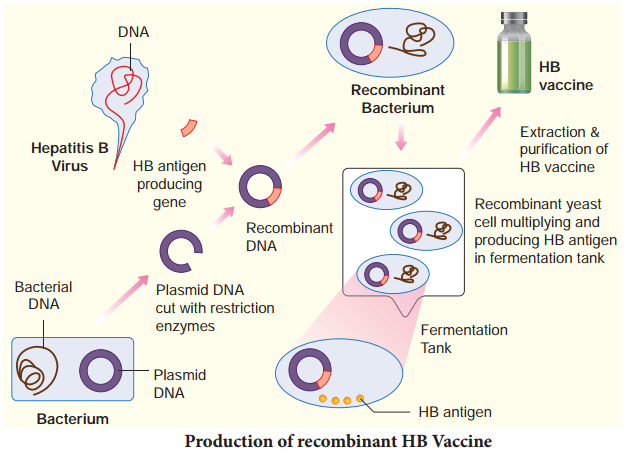
Recombinant hepatitis B vaccine as a subunit vaccine is produced by cloning hepatitis B surface antigen (HbsAg) gene in the yeast, Saccharomyces cerevisiae (Fig. 9.2).