Find free online Chemistry Topics covering a broad range of concepts from research institutes around the world.
Classification of Alcohols
Alcohols can be classified based on the number of hydroxyl groups and the nature of the carbon to which the functional group (- OH) is attached.
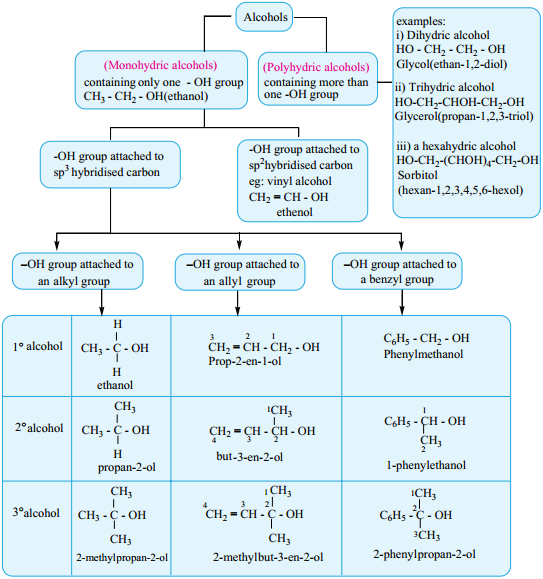
Alcohols may be classified as primary, secondary, or tertiary, according to which carbon of the alkyl group is bonded to the hydroxyl group. Most alcohols are colourless liquids or solids at room temperature.
Types of Alcohols:
Primary Alcohols:
A primary alcohol is one in which the hydroxyl group (- OH) is attached to a carbon atom with at least two hydrogen atoms.
Secondary Alcohols:
A secondary alcohol is one in which the hydroxyl group (- OH) is attached to a carbon with only one hydrogen atom attached.
Tertiary Alcohols:
One way of classifying alcohols is based on which carbon atom is bonded to the hydroxyl group. If this carbon is primary (1°, bonded to only one other carbon atom), the compound is a primary alcohol. A secondary alcohol has the hydroxyl group on a secondary (2°) carbon atom, which is bonded to two other carbon atoms.
The four types of alcohol are ethyl, denatured, isopropyl and rubbing. The one that we know and love the best is ethyl alcohol, also called ethanol or grain alcohol. It’s made by fermenting sugar and yeast, and is used in beer, wine, and liquor. Ethyl alcohol is also produced synthetically.
2 Types of Alcohols:
Distilled and Undistilled Alcohol:
There are two categories of alcoholic beverages:
Distilled and Undistilled.
The functional group in the alcohols is the hydroxyl group, – OH.
Primary alcohols are those alcohols where the carbon atom of the hydroxyl group (OH) is attached to only one single alkyl group. Some examples of these primary alcohols include Methanol (propanol), ethanol, etc.
Most believe the word “alcohol” originated in the Middle East since the prefix al is a definite article in Arabic the debate is about which word it stems from, either alcohol. “Alcohol” was later used specifically to mean ethanol, with the essence or spirit released through the distillation process.