Value Based Questions in Science for Class 10 Chapter 3 Metals and Non-metals
These Solutions are part of Value Based Questions in Science for Class 10. Here we have given Value Based Questions in Science for Class 10 Chapter 3 Metals and Non-metals
Question 1.
A customer approached a newly trained Goldsmith and asked him to make some ornaments. For this he gave advance money also. The Goldsmith bought a bar of 24-carat gold from the market and tried to make ornaments from it. He was not successful because the gold was too soft to handle. He approached a trained Goldsmith who asked him to use 22-carat gold for this purpose. He was now quite successful and could make the ornaments ?
- What is the difference between 24-carat gold and 22-carat gold ?
- What was wrong with 24-carat gold ?
- How did trained Goldsmith help the freshly trained Goldsmith ?
- What is the value associated with this episode ?
Answer:
- 24-carat gold is pure gold while 22-carat gold is comparatively less pure.
- Pure or 24-carat gold is very soft, little malleable and ductile. It is quite difficult to work on it. But 22 carat gold (alloy) is comparatively hard, more malleable and ductile
- Freshly trained Goldsmith realised his mistake and could complete the order. Otherwise he might have been in problem.
- The trained Goldsmith had a big heart and did not bother about the professional jealousy. He was very sincere in his approach and helped the junior colleage.
More Resources
- Value Based Questions in Science for Class 10
- HOTS Questions for Class 10 Science
- NCERT Solutions for Class 10 Science
- NCERT Exemplar Solutions for Class 10 Science
- Previous Year Question Papers for CBSE Class 10 Science
Question 2.
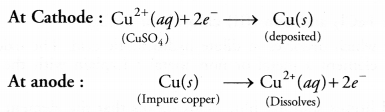
A student was asked to purify a sample of copper extracted by a suitable method. He constructed a-cell in which a rod of impure copper was made cathode while that of pure copper as anode. The electrolyte was aqueous CuSO4 solution. On passing electric current nothing happened. He realised his mistake and connected the electrodes in the reverse order. He was now quite successful in his mission.
- What was his mistake ?
- How did he rectify it ?
- Write a chemical equation for the reaction.
Answer:
- Impure copper cannot be made cathode and pure copper as the anode.
- He changed over. Pure copper was made cathode while impure copper as the anode.
- The chemical equations for the process are :
Hope given Value Based Questions in Science for Class 10 Chapter 3 Metals and Non-metals are helpful to complete your science homework.
If you have any doubts, please comment below. Learn Insta try to provide online science tutoring for you.